Assay QC states can be defined for use in approval workflows. Administrators define the states and can enable using QC states on assays which support them, including customized general purpose assays (GPAT). Admins also assign the QC Analyst role to any users who will be setting these states.
This topic covers the administrator tasks associated with Assay QC States. Learn about using the states in the topic:
Assay QC States: User Guide.
Do not use Assay QC in a
study folder. The use of QC states in studies for dataset QC may conflict with use for assays. To combine Assay QC and study functionality, define assays and studies in separate folders.
Assign the QC Analyst Role
In order to assign Assay QC states, users need the
QC Analyst role, which is assigned in the project or folder where it applies. Note that the QC Analyst role does not include Reader (or other) permissions automatically. Users must also have at least the Reader role in the folder.
- Select > Folder > Permissions.
- Add the users or groups to the QC Analyst role.
- Confirm that the users also have at least the Reader role in addition to the QC Analyst role.
- Click Save and Finish.
Enable Assay QC
You must enable the use of
QC States on at least one assay in a container in order to define or use states. You can enable this option during assay creation, or add it to an existing assay design created using the general purpose type.
- Open the assay designer:
- To start creating a new assay design, select > Manage Assays > New Assay Design, choose the "General" type and folder location, and click Next.
- Viewing an existing assay, select Manage Assay Design > Edit assay design.
- In the General Assay Designer, Assay Properties section, check the box for QC States.

- Click Save when finished.
Define Assay QC States
The assay design will now have an additional
QC State option on the grid menu. An Admin can define the desired QC states for the container by selecting
QC State > Manage states. Note that QC Analysts will not be able to see this option on the menu. Also note that the QC States listed here include all of the
dataset and assay QC states defined in the same folder.
- Viewing the assay runs grid, select QC State > Manage states.

- Click the green Add State icon to define the states your workflow requires. For example, you might define "Not Yet Reviewed", "Reviewed - Passed", and "Reviewed - Failed" for a simple workflow.

- For each state, enter:
- State Name: This name will be presented on the menu for users setting states.
- State Description: For your reference. This description is not displayed for the user.
- Public data: Checking this box means that the data in this state will be visible to data readers (i.e., possessors of the Reader role). For example, to hide runs that failed QC, you would uncheck this box for the 'Reviewed - Rejected' state.
- The In Use column indicates whether the state is in use for any assays in the folder.
- If you make a mistake, you can delete any row by clicking the red on the right.
- After defining the states you will need, click Save.
QC Analysts can see all the assay data in a folder, even that which has been assigned to QC states that are not "public", i.e. are not visible to non-analyst readers in the container.
Manage QC States
Once states are defined, when you reopen the
QC State > Manage states menu you will be able to set a default and make other changes.
Set a Default
There is a predefined "[none]" state that is the default default, but in the
Default states for assay data section you can select the state that newly imported data will have. Note that you will not be able to set a default QC state until after you have saved the set of states.
- From the assay runs grid, select QC State > Manage states.
- Under Default states for assay data, select the desired state from the set defined.
- Note that the dropdown list will only included states previously saved. If you add a new state, it will not appear on this list until you have saved and reopened this page.

In Use Status
The
In Use status column is set true when any runs in the container are set to that specific state. The "[none]" built in state cannot be deleted, so is always marked as "in use" even if the folder does not contain any runs, or contains only runs set to other states.
Delete Unused States
Cleaning up an outdated set of no longer in use states, or changing the set available can be done by deleting any states not in use. You can delete states individually or all at once.
- From the assay runs grid, select QC State > Manage states.
- Click the red on the right to delete any row.
- If you are deleting a row that has been saved, you will be asked to confirm the action.
- If you are deleting rows before you have clicked Save, no confirmation will be needed.
- Click Delete Unused QC States to delete all states not currently in use. You will be asked to confirm this action.
Do not delete Assay QC states in a
study folder. States that appear unused for Assay QC may be in use for dataset QC.
To combine Assay QC and study functionality, define assays and studies in separate folders.
Require Comments for QC State Changes
To require that users supply a comment whenever the QC state of a run is changed, select
Yes under
QC State Comments. This setting applies to all assays using QC states in the current container.
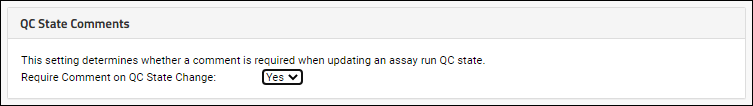
This will make the comment field required when a user changes changes the QC state.
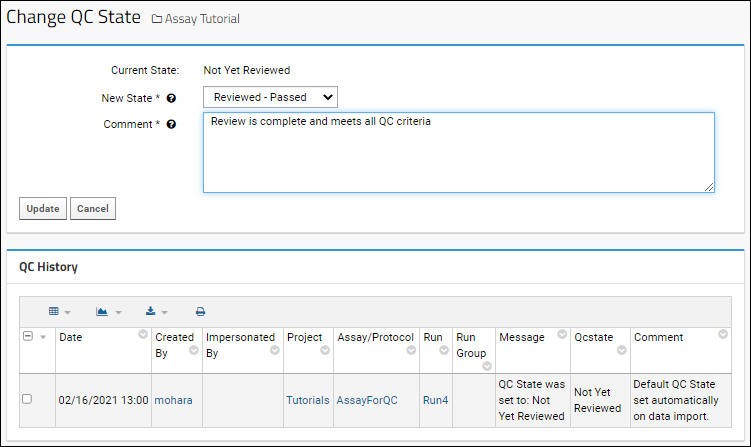
To see the history of state changes and comments for a given run, click the value in the
QC Flags column. The
QC History of all changes is shown below the change panel.
Related Topics