Overview
For large-scale studies and research projects, success depends on:
- Integrating many different kinds of information, including clinical, experimental, and sample data
- Tracking progress of the protocol and associated research projects
- Presenting your results in a secure way to colleagues
LabKey Study tools are designed around the specific requirements of longitudinal and cohort study management. Nearly every aspect of a study can be customized, including what data is captured by the system, the reports generated, and how the results are presented.
To get started, we provide two example tutorials:
Study Components
Participants
The default name for the individuals being studied is "participant". You can choose an alternate word to better match your working environment, such as "subject" or "patient", or can name the organism being studied, such as, "mouse", "mosquito", "tree", etc.
ParticipantIDs must be no longer than 32 characters.
Time
Tracking how various attributes of your participants vary over time, or more generally, what events happen in what sequence, is a typical requirement of study research. LabKey Server provides three different ways of measuring time in your study.
- Dates means that the time is broken into periods, called timepoints, bounded by calendar date, meaning that the amount of time elapsed between events is significant. The size of the timepoints can be customized so that months, weeks, years, or any other size 'blocks' become the units of time measurement.
- Assigned Visits means that the data is divided into named "events" in a sequence, possibly but not necessarily corresponding to an individual visiting a location. The actual dates may not be relevant, only the sequence in which they occur. For instance, "enrollment", "first vaccination", "second screening" might be named visits in a study. In a visit based study, data collection events are assigned a "sequence number", possibly but not necessarily using date information provided.
- Continuous is intended for open-ended observational studies that have no determinate end date, and no strong concept of dividing time into fixed periods. This style is useful for legacy or electronic health record (EHR) data.
Data
LabKey's study tools support the wide diversity of research data. Different data formats, methods of collection, analysis, and integration are brought together with convenient tools to support a wide variety of research efforts.
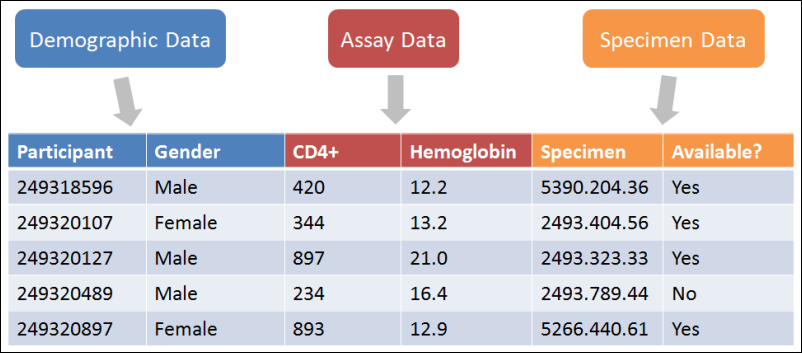
The datasets in a study repository come in three different types:
- Demographic.
- One row per participant.
- Demographic datasets record permanent characteristics of the participants which are collected only once for a study. Characteristics like birthplace, birth date, and enrollment date will not change over time.
- From a database point of view, demographic datasets have one primary key: the participantId.
- Each study needs at least one demographic dataset identifying the participants in a study.
- Clinical.
- One row per participant/timepoint pair.
- Clinical datasets record participant characteristics that vary over time in the study, such as physical exam data and simple lab test data. Typical data includes weight, blood pressure, or lymphocyte counts. This data is collected at multiple times over the course of the study.
- From a database point of view, clinical datasets have two primary keys: the participantId and a timepoint.
- Assay/Instrument.
- Multiple rows per participant/timepoint are allowed.
- These datasets record the assay or instrument data in the study. Not only is this data typically collected repeatedly over time, but more than one of each per timepoint is possible, if, for example, multiple vials of blood are tested, or multiple dilutions of a product are tested on one sample.
- From a database point of view, assay datasets have at least two, and possibly three keys: participant ID and timepoint, plus an optional third key such as a sampleID.
Integrate
LabKey Server knows how to integrate the
diverse range of research data, including demographic, clinical, experimental, and sample data.
Track Progress
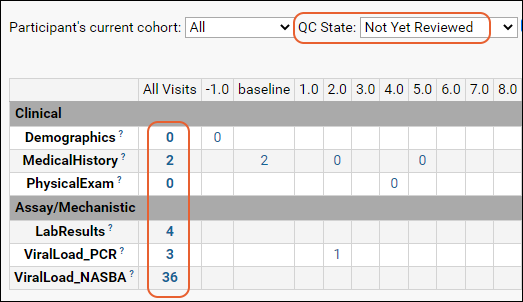
Progress tracking tools help you put all the pieces together.
Present Results
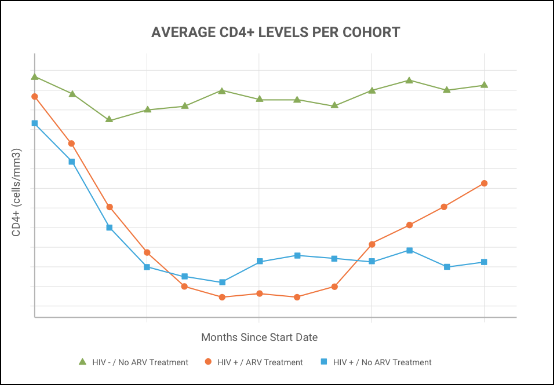
Present your research using LabKey's rich
analytic and visualization tools.
Learn More
Use the following topics to get started: