This topic describes options for managing assay designs. Assay designs can be defined in a local container, or shared from a project or sitewide from the
Shared project. Be aware that editing a design shared in other locations will change it for all users. Another alternative is to edit a copy of the design.
Manage an Assay Design
Open the list of currently defined assays by selecting
> Manage Assays. Click on the name of any assay to open the runs page. The
Manage Assay Design menu provides the following options:

- Edit assay design: Add, delete, or change properties or structure.
- The Name is always required.
- For Standard assay designs the Name can be edited after creation, but must remain unique. For Specialty assay designs, editing the Name after creation is not allowed.
- Note that all current users of the assay design, including those in subfolders, will be impacted by these changes.
- Copy assay design: This option lets you create a new assay design based on the current one.
- Changes to the copy do not affect the original design or runs that use it.
- You can create the copy in the same location or choose another folder. Note that in a Biologics or Sample Manager project, assays can only be created at the project level.
- Delete assay design: This option will delete the current design as well as all runs that used it. You will see a list of what would be deleted and have a chance to confirm or cancel this action.
- Export assay design: The design will be exported to a XAR file you can import elsewhere. See Export/Import Assay Design.
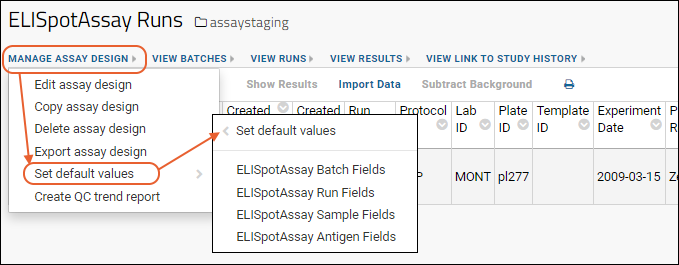
- Set default values: Described below.
- (Premium feature) Create QC trend report: Create a QC trend report to track metrics relevant to your instrument. Learn more in this topic: Quality Control Trend Reports.
Set Default Values
Fields in an assay design can support defining default values using the
field editor. Defaults can be of several types: last entered, editable default, and a fixed value.
Values for editable and fixed value properties are then set using the
Set Default Values option on the
Manage Assay Design menu. The assay design may be then be inherited in subfolders, which may override these parent defaults if needed using the
Set Default Values option in the other container. These folder defaults will, in turn, be inherited by sub-folders that do not specify their own defaults.
You can set defaults for:
- Batch fields
- Run fields
- Properties specific to the assay type. For example, for an Luminex assay, additional items would include "analyte" and "Excel run file" properties.
Assay Data Auditing and Tracking Changes
Some assays, like the Standard assay type, allow you to make run and data rows editable individually. Editability at the run or result level is enabled in the assay design by an administrator. Any edits are audited, with values before and after the change being captured. See
Assay/Experiment events in the
audit log. Upon deleting assay data, the audit log records that a deletion has occurred, but does not record what data was deleted.
Some assay types, including Standard and Luminex, allow you to upload a
replacement copy of a file/run. This process is called "re-import" of assay data. The server retains the previous copy and the new one, allowing you to review any differences.
See the
Assay Feature Matrix for details about which assay types support editable runs/results and re-import.
Related Topics
